 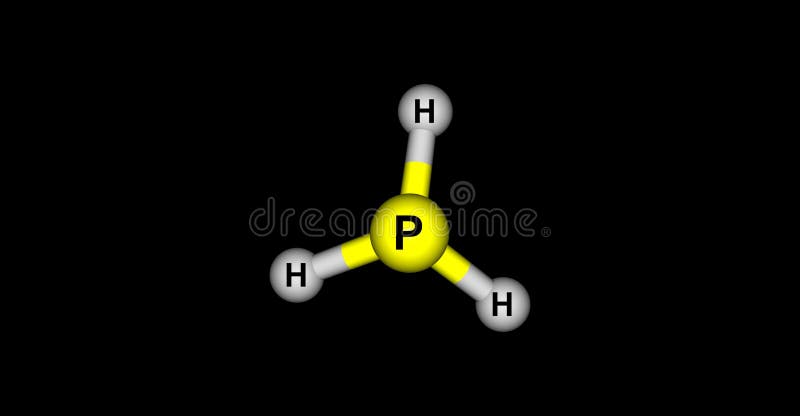
To know the Lewis Structure, we first know the central atom and the arrangement of other atoms. PH3 Lewis Structure Lewis Structure is the pictorial representation of the arrangement of atoms and valence electrons in the molecule. In phosphine, orthogonal 3p orbitals overlap with 1s orbital of H. 8 There are eight valence electrons for the Phosphine or PH3 molecule. 5 degrees betweenhydrogen-phosphorus-hydrogen bonds. 93.7 H P H 9.5 Hybrid Orbitals and Molecular Geometry There are many. Reasons: The pure p orbitals take part in bonding. Phosphine or PH 3 has a molecular structure of a trigonal pyramidal having a bond angle of 93. H PRACTICE EXERCISE 9.11 The phosphine molecule, PH3, has a trigonal pyramidal. Hence, the molecular geometry of PH3 is trigonal pyramidal but there is no hybridization due to PH3 being a Drago molecule. Trending Questions What is the direction of rotation of earth at the north pole? What is the large red twinkling star seen in the south east? If the earth stopped spinning would you lose gravity? What day of the week was 29th September 1958? How can you tell that the Earth has a magnetic field? Does mass measurement include the effect of earth's gravitational pull on the object being measured? What is the time interval between vernal equinoxes called? The Northern hemisphere experiences winter when Earth is farthest from the sun True or False? What degree is the north star? At what phase are the tides least noticeable? What does the Moon enter when a partial lunar eclipse begins? If two stars differ by 8. In essence, PH3 is a Drago molecule and thus its bond angle shows that the p-orbitals have an angle of 90.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
